HEPATITIS B CAN POSE A SERIOUS THREAT
Without protection, an acute hepatitis B infection may develop into an incurable, contagious, and potentially deadly chronic infection1
In the United States alone:
- ~22,000 acute* hepatitis B cases2
- Up to 2.2 million people living with chronic hepatitis B3†
- About 4000 people die from chronic hepatitis B–related cirrhosis and 1500 die from hepatocellular carcinoma each year4

HBV=hepatitis B virus; HBsAg=hepatitis B surface antigen.
*Defined as acute illness with discrete onset of symptoms and jaundice or elevated serum alanine aminotransferase (ALT) >100 IU/L.2
†Defined as unable to clear the hepatitis B virus after 6 months.1
‡Based on the 2015 Centers for Disease Control and Prevention Viral Hepatitis Surveillance report.
Hepatitis B seroprotection remains a serious issue
- The HBV vaccine series alone takes up to 2 weeks to achieve initial serum levels and 3 doses (6 months) to provide seroprotection in ~90% of patients7-9
- Waning serum antibody levels may compromise seroprotection over time
– Among immunocompetent HBV vaccine responders, protection lasts 15 to 20 years7
- Response can be compromised due to smoking history, obesity, age, chronic medical conditions, compromised immune system, or gender (male)7


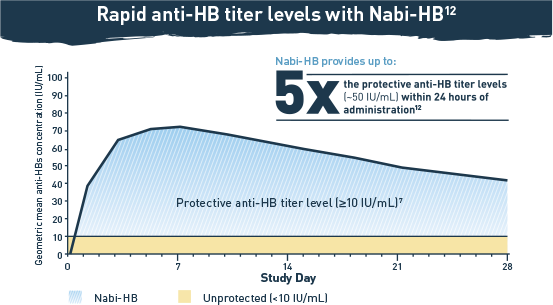
Nabi-HB® PROVIDES PROTECTION AGAINST HEPATITIS B INFECTION WITHIN 24 HOURS OF ADMINISTRATION11,12
Guaranteed potency with Nabi-HB11
- Each milliliter of Nabi-HB contains >312 IU/mL of anti-HBs
- The potency of each milliliter of Nabi-HB exceeds the potency of anti-HBs in a US reference hepatitis B immune globulin (FDA). The US reference has been tested against the WHO standard and found to be equal to 208 IU/mL
Anti-HBs=anti-hepatitis B surface antibodies; IU=international units; WHO=World Health Organization.
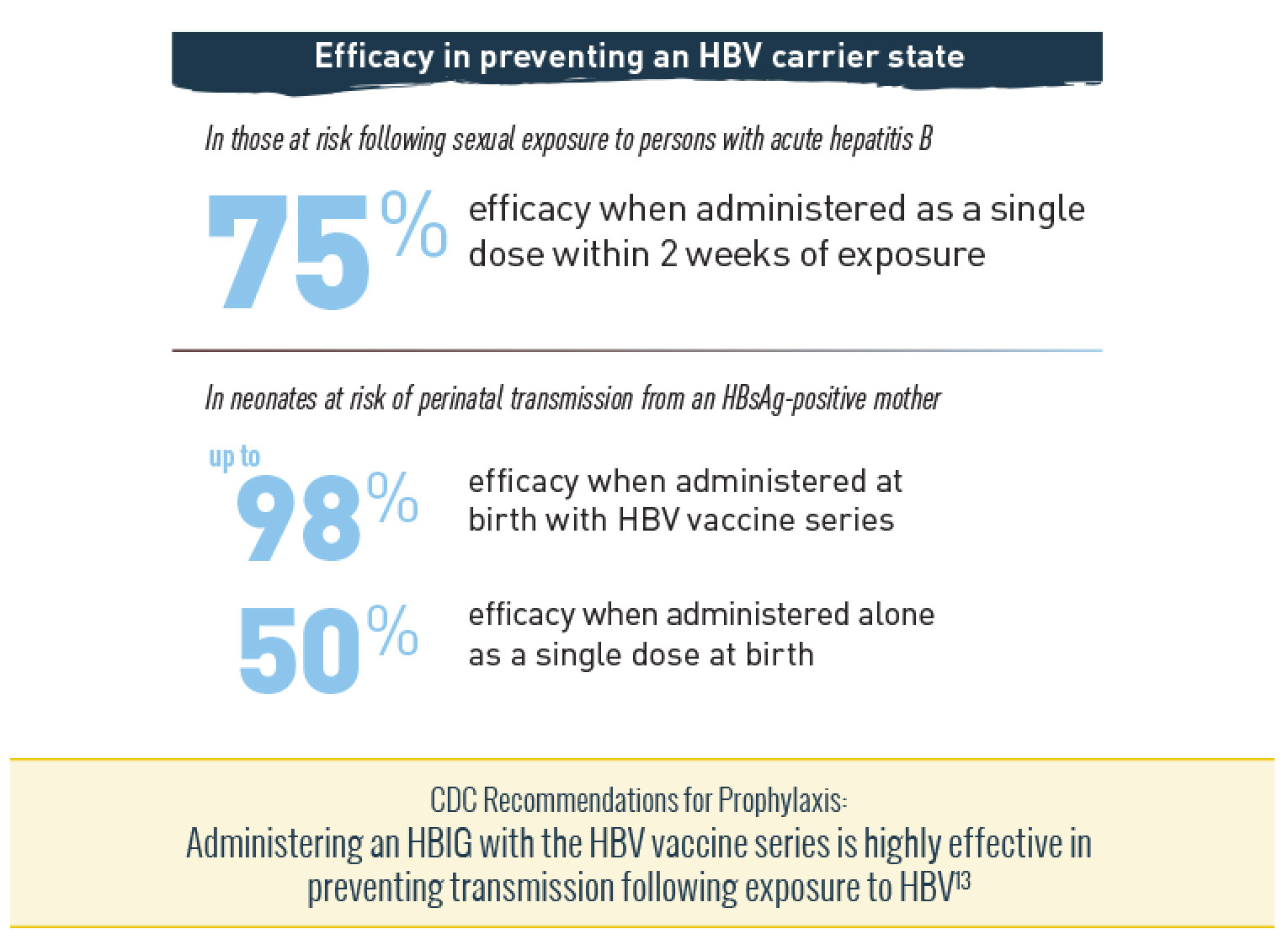
Nabi-HB® + HBV VACCINE SERIES DELIVERS HIGHLY EFFECTIVE PROTECTION11
HBIG=hepatitis immunoglobulin.
Nabi-HB® WAS WELL TOLERATED AND REACTIONS REPORTED WERE MILD
Nabi-HB® IS DEVELOPED THROUGH A SAFETY-FIRST MANUFACTURING PROCESS
- Manufactured solely from US plasma from the Food and Drug Administration (FDA)-licensed plasma collection centers
- Produced using a manufacturing process based on cold ethanol fractionation followed by ion exchange chromatography
Three virus inactivation/removal steps
- Precipitation and removal of fraction III of the cold ethanol process removes some nonenveloped viruses
- Solvent/detergent treatment designed to inactivate enveloped viruses
- Nanofiltration with 35 nm filters to remove nonenveloped and enveloped viruses
Viral safety built into manufacturing
- Multistep viral removal/activation system that eliminates and inactivates viruses to further ensure the safety of Nabi-HB
– Because this product is made from human blood, it may carry a risk of transmitting infectious agents, eg, viruses, the variant Creutzfeldt-Jakob disease (vCJD) agent and, theoretically, the Creutzfeldt-Jakob disease (CJD) agent
Nabi-HB is a market leader with a proven safety profile11,12
References:
- World Health Organization. Hepatitis B fact sheet: http://www.who.int/mediacentre/factsheets/fs204/en/. Accessed January 18, 2018.
- Centers for Disease Control and Prevention. Viral Hepatitis Surveillance Report, United States, 2015. https://www.cdc.gov/hepatitis/statistics/2015surveillance/pdfs/2015HepSurveillanceRpt.pdf+&cd=3&hl=en&ct=clnk&gl=us. Accessed January 18, 2018.
- Centers for Disease Control and Prevention. Hepatitis B FAQs for the Public. https://www.cdc.gov/hepatitis/hbv/bfaq.htm. Accessed January 11, 2018.
- Centers for Disease Control and Prevention. Epidemiology and prevention of vaccine-preventable disease: hepatitis B. https://www.cdc.gov/vaccines/pubs/pinkbook/hepb.html. Accessed January 18, 2018.
- Centers for Disease Control and Prevention. Protect your baby for life: when a pregnant woman has hepatitis B. https://www.cdc.gov/hepatitis/hbv/pdfs/hepbperinatal-protectwhenpregnant.pdf. Updated October 2010. Accessed January 18, 2018.
- Centers for Disease Control and Prevention. Sharps injuries: bloodborne pathogens. https://www.cdc.gov/niosh/stopsticks/bloodborne.html. Accessed January 18, 2018.
- Centers for Disease Control and Prevention. A comprehensive immunization strategy to eliminate transmission of hepatitis B virus infection in the United States: recommendations of the Advisory Committee on Immunization Practices (ACIP) part 2: immunization of infants, children, and adolescents. MMWR Recomm Rep. 2006;55(RR-16):1-40.
- Junewicz A, Brateanu A, Nielsen C. Q: do patients who received only two doses of hepatitis B vaccine need a booster? Cleve Clin J Med. 2014;81(6):346-348.
- PDR: prescriber’s digital reference. Engerix (hepatitis B vaccine recombinant) drug summary: http://www.pdr.net/drug-summary/Engerix-Bhepatitis-B-vaccine—recombinant—186.5915. Accessed January 17, 2018.
- Centers for Disease Control and Prevention (CDC). Adult vaccination coverage—United States, 2010. MMWR Morb Mortal Wkly Rep. 2012;61(4):66-72
- Nabi-HB [Prescribing Information]. Boca Raton, FL: Biotest Pharmaceuticals Corporation; 2008.
- Data on file. ADMA Biologics.
- Workowski KA, Bolan GA; Centers for Disease Control and Prevention. Sexually transmitted diseases treatment guidelines, 2015. MMWR Recomm Rep. 2015;64:1-137.